Change Location
You are about to enter the PAJUNK® GmbH
Medizintechnologie (global) website.
If you are from United States,
visit the U. S. Website:
 pajunkusa.com
pajunkusa.com
You are about to enter the PAJUNK® GmbH
Medizintechnologie (global) website.
If you are from United States,
visit the U. S. Website:
 pajunkusa.com
pajunkusa.com
You are about to enter the PAJUNK® GmbH
Medizintechnologie (global) website.
If you are from United Kingdom, Ireland, Malta or India,
visit the UK Website:
 pajunk.co.uk
pajunk.co.uk
Together with Pajunk, Prof. Sprotte developed the Sprotte, the first atraumatic needle for lumbar Puncture. The secret of its success can be found in its unique tip geometry and basic architecture. This design, developed especially for the requirements of dural puncture, allows for an atraumatic puncture of the ligamentary structures and optimises CSF flow while reducing the incidence of post-lumbar puncture headaches (PLPH). Sprotte decreases complications of Lumbar Puncture and increases the safety of application, and the efficiency of diagnostics.
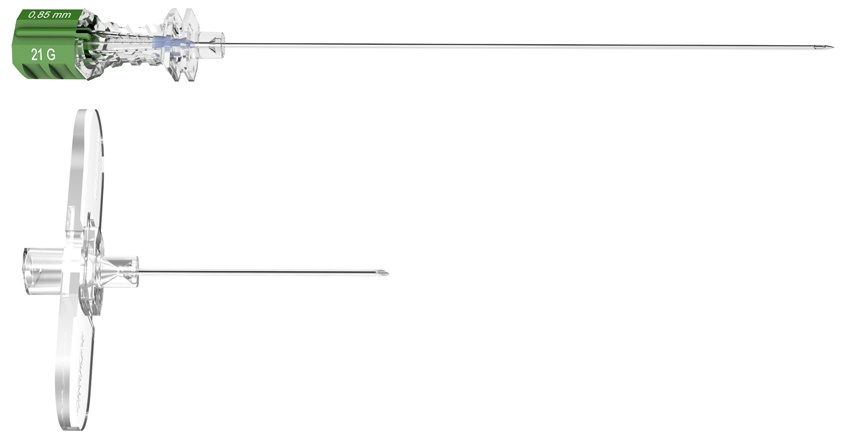









| Item description | Item no. LUER |
Item no.
|
Purchase Unit |
|---|---|---|---|
|
22G x 90mm (3 1/2") |
001151-30C | 25 | |
|
22G x 120mm (4 3/4") |
031151-30C | 031163-30C | 10 |
|
21G x 90mm (3 1/2") |
001151-31A | 001163-31A | 25 |
|
20G x 90mm (3 1/2") |
0001151-31 | 0001163-31 | 25 |
|
22G x 90mm (3 1/2") with standard hub, NRFit with magnifying hub |
001163-30C | 25 |
| Item description |
22G x 90mm (3 1/2") |
|
|---|---|---|
| Item no. LUER | 001151-30C | 25 |
| Item description |
22G x 120mm (4 3/4") |
|
| Item no. LUER | 031151-30C | 10 |
| Item no. NRFIT | 031163-30C | 10 |
| Item description |
21G x 90mm (3 1/2") |
|
| Item no. LUER | 001151-31A | 25 |
| Item no. NRFIT | 001163-31A | 25 |
| Item description |
20G x 90mm (3 1/2") |
|
| Item no. LUER | 0001151-31 | 25 |
| Item no. NRFIT | 0001163-31 | 25 |
| Item description |
22G x 90mm (3 1/2") with standard hub, NRFit with magnifying hub |
|
| Item no. LUER | 25 | |
| Item no. NRFIT | 001163-30C | 25 |
| Item description | Item no. LUER |
Item no.
|
Purchase Unit |
|---|---|---|---|
|
22G x 90mm (3 1/2") with wings |
321151-30C | 321163-30C | 25 |
|
21G x 90mm (3 1/2") with wings |
321151-31A | 321163-31A | 25 |
|
20G x 90mm (3 1/2") with wings |
331151-31B | 331163-31B | 25 |
| Item description |
22G x 90mm (3 1/2") with wings |
|
|---|---|---|
| Item no. LUER | 321151-30C | 25 |
| Item no. NRFIT | 321163-30C | 25 |
| Item description |
21G x 90mm (3 1/2") with wings |
|
| Item no. LUER | 321151-31A | 25 |
| Item no. NRFIT | 321163-31A | 25 |
| Item description |
20G x 90mm (3 1/2") with wings |
|
| Item no. LUER | 331151-31B | 25 |
| Item no. NRFIT | 331163-31B | 25 |
| Item description | Item no. LUER |
Item no.
|
Purchase Unit |
|---|---|---|---|
|
22G x 103mm (4") with wings |
341151-30C | 25 | |
|
22G x 120mm (4 3/4") with wings |
131151-30C | 10 | |
|
21G x 103mm (4") with wings |
341151-31A | 341163-31A | 25 |
|
21G x 120mm (4 3/4") with wings |
331151-31A | 331163-31A | 25 |
|
20G x 120mm (4 3/4") with wings |
321151-31B | 321163-31B | 25 |
|
20 G x 150mm with wings |
361151-31B | 10 |
| Item description |
22G x 103mm (4") with wings |
|
|---|---|---|
| Item no. LUER | 341151-30C | 25 |
| Item description |
22G x 120mm (4 3/4") with wings |
|
| Item no. LUER | 131151-30C | 10 |
| Item description |
21G x 103mm (4") with wings |
|
| Item no. LUER | 341151-31A | 25 |
| Item no. NRFIT | 341163-31A | 25 |
| Item description |
21G x 120mm (4 3/4") with wings |
|
| Item no. LUER | 331151-31A | 25 |
| Item no. NRFIT | 331163-31A | 25 |
| Item description |
20G x 120mm (4 3/4") with wings |
|
| Item no. LUER | 321151-31B | 25 |
| Item no. NRFIT | 321163-31B | 25 |
| Item description |
20 G x 150mm with wings |
|
| Item no. LUER | 361151-31B | 10 |
| Item description | Item no. LUER |
Item no.
|
Purchase Unit |
|---|---|---|---|
|
Ø 1,2 x 30mm (1 1/5") for 20G |
061151-30L | 061163-30L | 25 |
|
Ø 1.0 x 30 mm (1 1/5") for 22G |
001151-30L | 001163-30L | 25 |
| Item description |
Ø 1,2 x 30mm (1 1/5") for 20G |
|
|---|---|---|
| Item no. LUER | 061151-30L | 25 |
| Item no. NRFIT | 061163-30L | 25 |
| Item description |
Ø 1.0 x 30 mm (1 1/5") for 22G |
|
| Item no. LUER | 001151-30L | 25 |
| Item no. NRFIT | 001163-30L | 25 |
| Downloads |
|---|
Sprotte Lumbar Brochure
|
Studies:
1 Davis, A.; Dobson, R.; Kaninia, S.; Giovannoni, G.; Schmierer, K. (2016): Atraumatic needles for lumbar puncture: why haven't neurologists changed? In Practical neurology 16 (1), pp. 1822. DOI: 10.1136/practneurol-2014-001055.02 Engedal, Thorbjørn S.; Ørding, Helle; Vilholm, Ole Jakob (2015): Changing the needle for lumbar punctures: results from a prospective study. In Clinical neurology and neurosurgery 130, pp. 7479. DOI: 10.1016/j.clineuro.2014.12.020.3 Tung, C. E.; Yuen, T. S.; Lansberg, M. G. (2012): Cost comparison between the atraumatic and cutting lumbar puncture needles. In Neurology 78, pp. 109113.4 Arevalo-Rodriguez, Ingrid; Muñoz, Luis; Godoy-Casasbuenas, Natalia; Ciapponi, Agustín; Arevalo, Jimmy J.; Boogaard, Sabine; Roqué I Figuls, Marta (2017): Needle gauge and tip designs for preventing post-dural puncture headache (PDPH). In The Cochrane database of systematic reviews 4, CD010807. DOI: 10.1002/14651858.CD010807.pub2.5 McLaughlin, Colleen A.; Hockenberry, Marilyn J.; Kurtzberg, Joanne; Hueckel, Rémi; Martin, Paul L.; Docherty, Sharron L. (2014): Standardization of health care provider competencies for intrathecal access procedures. In Journal of pediatric oncology nursing: official journal of the Association of Pediatric Oncology Nurses 31 (6), pp. 304316. DOI: 10.1177/1043454214543019.6 Nath, Siddharth; Koziarz, Alex; Badhiwala, Jetan H.; Alhazzani, Waleed; Jaeschke, Roman; Sharma, Sunjay et al. (2018): Atraumatic versus conventional lumbar puncture needles: a systematic review and meta-analysis. In The Lancet 391 (10126), pp. 11971204. DOI: 10.1016/S0140-6736(17)32451-0.7 Rochwerg, Bram; Almenawer, Saleh A.; Siemieniuk, Reed A. C.; Vandvik, Per Olav; Agoritsas, Thomas; Lytvyn, Lyubov et al. (2018): Atraumatic (pencil-point) versus conventional needles for lumbar puncture: a clinical practice guideline. In BMJ, k1920. DOI: 10.1136/bmj.k1920.8 Zhang, Yi C.; Chandler, Alexander J.; Kagetsu, Nolan J. (2014): Technical compliance to standard guidelines for lumbar puncture and myelography: survey of academic neuroradiology attendings and fellows. In Academic radiology 21 (5), pp. 612616. DOI: 10.1016/j.acra.2014.01.021.9 Bellamkonda, Venkatesh R.; Wright, Thomas C.; Lohse, Christine M.; Keaveny, Virginia R.; Funk, Eric C.; Olson, Michael D.; Laack, Torrey A. (2017): Effect of spinal needle characteristics on measurement of spinal canal opening pressure. In The American journal of emergency medicine 35 (5), pp. 769772. DOI: 10.1016/j.ajem.2017.01.047.10 Puolakka, R.; Andersson, L. C.; Rosenberg, H. (2000): Microscopic Analysis of Threee Different Spinal Needle Tips After Experimental Subarachnoid Puncture. In Regional Anesthesia and Pain Medicine 25 (2), pp. 163169.11 Lavi R., Rowe J.M., Avivi I. Lumbar Puncture. It Is Time to Change the Needle, Eur Neurol, 2010; 64:108113
NRFit® is a registered trademark of GEDSA and is used with their permission
