Change Location
You are about to enter the PAJUNK® GmbH
Medizintechnologie (global) website.
If you are from United States,
visit the U. S. Website:
 pajunkusa.com
pajunkusa.com
You are about to enter the PAJUNK® GmbH
Medizintechnologie (global) website.
If you are from United States,
visit the U. S. Website:
 pajunkusa.com
pajunkusa.com
You are about to enter the PAJUNK® GmbH
Medizintechnologie (global) website.
If you are from United Kingdom, Ireland, Malta or India,
visit the UK Website:
 pajunk.co.uk
pajunk.co.uk
Ultrasound-guided localisation of peripheral nerves and the associated real-time visualisation provide crucial benefits in regional anaesthesia.1 However, it is clear that this does not reduce the incidence of permanent nerve damage.2 Even in combination with nerve stimulation, intrafascicular injections cannot be ruled out.1,3,4 The causes include incorrect positioning of the needle along with exceeding critical injection pressures.
With the automatic injection pressure limiter NerveGuard, developed by Pajunk, the injection pressure is automatically limited to avoid nerve damage.


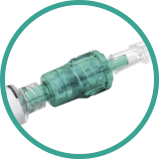
| Item description | Item no. LUER |
Item no.
|
Purchase Unit |
|---|---|---|---|
|
NerveGuard / single / sterile / For single shot applications in diameters of 20G/21G/22G |
001151-38M | 001163-38M | 10 |
| Item description |
NerveGuard / single / sterile / For single shot applications in diameters of 20G/21G/22G |
|
|---|---|---|
| Item no. LUER | 001151-38M | 10 |
| Item no. NRFIT | 001163-38M | 10 |
| Downloads |
|---|
Regional Anesthesia Catalogue Brochure
|
Studies:
1 Choquet O., Capdevila X. Ultrasound-guided nerve blocks: the real position of the needle should be defi ned, Anaesth. Analg. 2012 May; 114(5): 929–930
2 Neil J. M., Brull R., Horn J. L., Liu S. S., McCartney C. J., Perlas A., Salinas F. V., Tsui B. C. The Second American Society of Regional Anesthesia and Pain Medicine Evidence-Based Medicine Assessment of Ultrasound-Guided Regional Anesthe-sia, Reg. Anaesth. Pain Med. 2016 March–April; 41(2): 181–194
3 Robards C., Hadzic A., Somasundaram L., Iwata T., Gadsden J., Xu D., Sala-Blanch X. Intraneural injection with low-current stimulation during popliteal sciatic nerve block, Anesth. 2009 Aug; 109(2): 673– 677
4 Vassiliou T., Müller H. H., Limberg S., De Andres J., Steinfeldt T., Wiesmann T. Risk evaluation for needle-nerve contact related to electrical nerve stimulation in a porcine model, Acta Anaes-thesiol. Scand. 2016 Mar; 60(3): 400–406
6 Hadzic A., Dilberovic F., Shah S., Kulenovic A., Kapur E., Zaciragic A., Cosovic E., Vuckovic I., Divanovic K. A., Mornjakovic Z., Thys D. M., Santos A. C. Combination of intraneural injection and high injection pressure leads to fascicular injury and neurologic deficits in dogs, Reg. Anesth. Pain Med. 2004 September–October; 29(5): 417–423 8 Gadsden J., Choi J. J., Lin E., Robinson A. Opening injection prewssure consistently detects needle-nerve contact during ultrasound-guided interscalene brachial plexus block, Anesthesiology 2014 May; 120(5): 1246–1253
9 Gadsden J., Latmore M., Levine D. M., Robinson A. High Opening Injection Pressure is Associated With Needle-Nerve and Needle-Fascia Contact During Femoral Nerve Block, Reg. Anesth. Pain Med. 2016 Jan–Feb; 41(1): 50–55
10 Steinfeldt T., Graf J., Schneider J., Nimphius W., Weihe E., Borgeat A., Wulf H., Wiesmann T. Histological consequences of needle-nerve contact following nerve stimulation in a pig model, Anesth. Research and Practice 2011; Article ID 591851: 0–9 11 Lundborg G., Myers R., Powell, H. Nerve compression injury and increased endoneurial fluid pressure: a „miniature compartment syndrome“, J. Neurol. Neurosurg. Psychiatry 1983 Dec; 46(12): 1119–1124
NRFit® is a registered trademark of GEDSA and is used with their permission
